Common Challenges in E-Prescribing Integration
E-prescribing has transformed how healthcare providers manage prescriptions, reducing errors and improving safety.
Yet, integrating these systems into existing workflows is far from simple.
Key challenges include:
Interoperability Issues: Many EHR systems struggle to exchange data effectively due to incompatible technical standards and fragmented systems.
Workflow Disruptions: Poor system design can slow clinicians down, create inefficiencies, and increase errors.
Data Quality Problems: Errors in medication data, free-text entries, and unstructured notes lead to delays and safety risks.
Regulatory Compliance: Meeting strict DEA, CMS, and HIPAA requirements for controlled substances and patient data is complex.
Staff Resistance: Clinicians often resist new systems, citing usability issues and alert fatigue.Solutions include adopting standardized data protocols (e.g., FHIR, RxNorm), embedding e-prescribing features directly into EHRs, improving alert systems, and offering tailored training programs.
Platforms like Opus Behavioral Health EHR address these pain points by integrating advanced tools, ensuring compliance, and streamlining workflows.
E-Prescribing Integration Challenges: Key Statistics and Impact on Healthcare
What Drives EHR Innovation in Prescription Management and How Technology Can Help
Data Exchange and Interoperability Issues
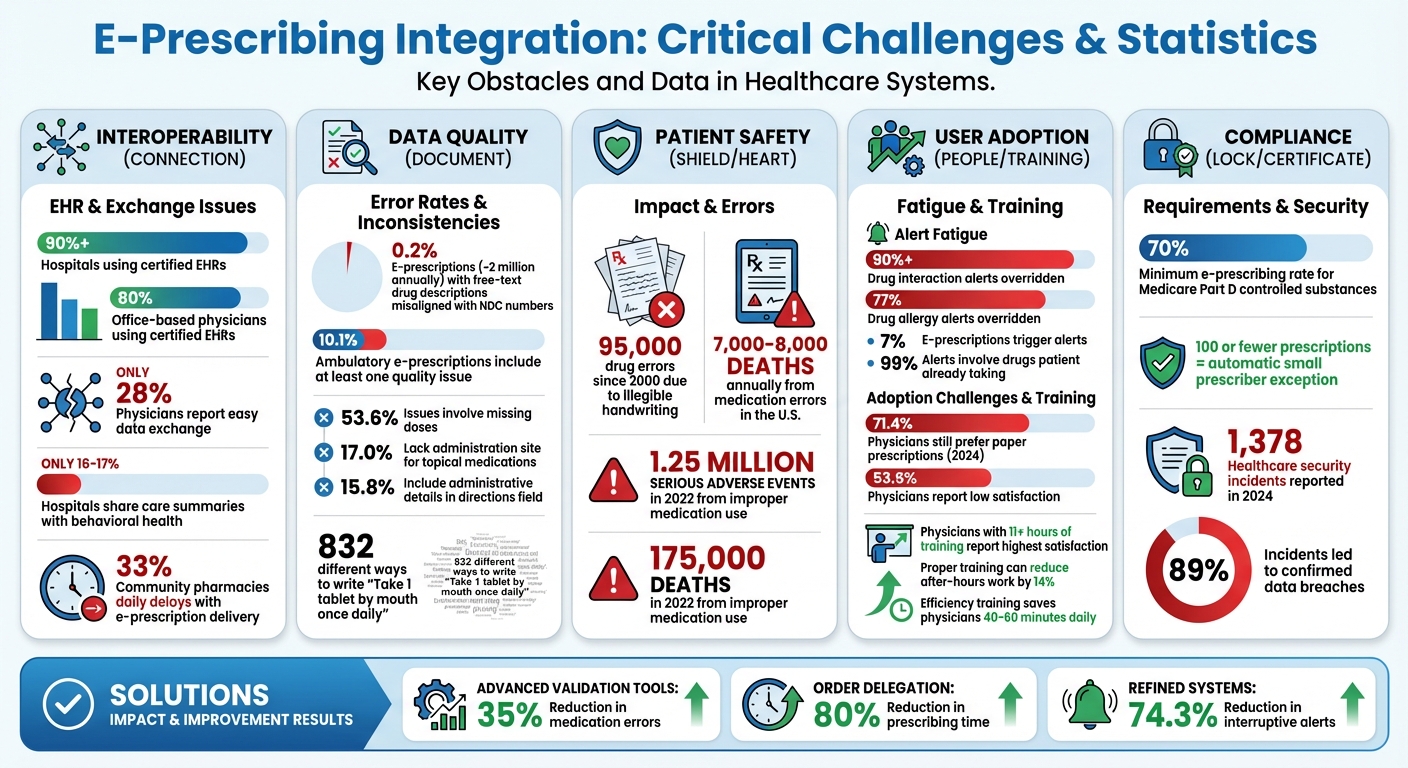
Even though over 90% of hospitals and 80% of office-based physicians in the U.S. use certified EHRs, many systems still struggle to exchange data effectively [11]. These technical limitations create significant hurdles, which are explored further in the following sections.
Incompatible Technical Standards
The healthcare industry relies on various data standards, but mismatches between these standards can cause major problems.
For instance, one hospital may use HL7 version 2, while another relies on FHIR, leading to communication gaps [9][11]. Similarly, inconsistent use of terminology systems like SNOMED CT, LOINC, and ICD can result in serious misunderstandings.
A striking example: over 0.2% of e-prescriptions (around 2 million annually) include free-text drug descriptions that misalign with National Drug Code (NDC) numbers, creating potential risks for patient safety [13].
"The inconsistent adoption of standards remains a persistent roadblock." - Brayan Vasquez, PCIS GOLD [9]
Isolated Systems and Fragmented Data
Data fragmentation remains a critical issue, forcing healthcare providers to make decisions without access to complete patient records.
For example, only 16-17% of hospitals share care summaries with behavioral health providers, leaving gaps in vital information [7].
This lack of communication is particularly harmful for behavioral health practices managing conditions like substance use disorders, where missing medication histories can lead to harmful drug interactions or duplicate prescriptions.
The problem extends beyond behavioral health. Only 28% of physicians report that sending and receiving patient data across different EHR platforms is easy [10].
Additionally, one-third of community pharmacies experience daily delays where patients arrive to pick up prescriptions before the electronic script has reached the pharmacy [12]. These inefficiencies not only waste time but also increase risks for patients.
How to Improve Interoperability
Starting January 1, 2026, ONC-certified EHRs will be required to support USCDI v3. Implementing FHIR-based APIs and middleware solutions can help bridge gaps by translating data across different systems in real time [7][9][11].
Organizations can also join Qualified Health Information Networks (QHINs) under the Trusted Exchange Framework and Common Agreement (TEFCA) to streamline data sharing [7][11].
To ensure data integrity during transfers, adopting unified terminology standards is key. Using SNOMED CT for diagnoses, LOINC for lab results, and RxNorm for medications can minimize errors and misinterpretations [9][11]. For behavioral health providers, updates to 42 CFR Part 2 - effective February 16, 2026 - allow single-consent for sharing substance use disorder records, removing a major legal barrier to data exchange [7].
Specialized platforms like Opus Behavioral Health EHR can also simplify interoperability by offering advanced middleware and real-time API integrations, making data exchange smoother and more reliable.
Integration and Workflow Compatibility
Even when e-prescribing systems meet compliance standards, they can still disrupt the flow of daily clinical tasks. Beyond just fixing technical issues, it’s crucial to align these systems with how healthcare teams actually work.
When software design doesn’t match the real-world routines of providers, nurses, and medical assistants, it creates inefficiencies and increases the likelihood of mistakes.
Here’s a closer look at how to address these challenges through better medication data management, workflow alignment, and user role management.
Medication Data Mapping
System mismatches often lead to issues at the pharmacy, like when a patient’s name appears as "Judy" in the prescriber’s EHR but as "Judith" in the pharmacy’s system.
These discrepancies contribute to nearly half of all medication errors, which rack up significant costs every year [14].
One effective solution is to use RxNorm as the standard drug identifier, as it avoids the inconsistencies found with National Drug Codes (NDCs), which differ by manufacturer [17].
Additionally, systems should prioritize structured "Sig" fields (patient directions) over free-text entries to reduce ambiguity. Another helpful safeguard is using distinctive lettering (e.g., buPROPion vs. busPIRone) to prevent confusion between look-alike or sound-alike medications in dropdown menus [8][17].
These measures are especially important for behavioral health providers prescribing controlled substances, where errors are more common. For instance, one study found a 7% error rate among Schedule III-V drugs in a sample of 2,000 e-scripts [16].
Adapting to Different Workflows
Switching between an EHR and a separate e-prescribing system can slow clinicians down and introduce errors [15].
This disconnect often results in pharmacy updates being overlooked or cancellations made in the EHR not reaching the pharmacy, leading to incorrect or duplicate prescriptions [1][8].
"If the software doesn't integrate cleanly with your EHR - if it's not easy, intuitive, and built for how hospice care actually happens - it's not going to fly." - BetterRX [15]
The best approach is embedding e-prescribing features directly into the patient’s electronic chart. For behavioral health providers, platforms like Opus Behavioral Health EHR offer seamless integration, keeping all prescribing tasks unified in one place.
Another time-saving feature is order delegation, which allows medical assistants or nurses to prepare routine refills for provider approval. This setup can cut prescribing time by as much as 80%, without sacrificing oversight [14].
Practices using these tools have reported a 35% drop in medication errors [14]. Additionally, tailoring system access to specific clinical roles can further streamline workflows and enhance security.
Managing User Access by Role
Role-specific access controls are key to protecting patient data and maintaining efficiency.
Role-Based Access Control (RBAC) assigns permissions based on job responsibilities - medical assistants can prepare prescriptions, but only licensed providers can sign and send them, particularly for controlled substances [14][6].
This delegation model eliminates the bottleneck of providers manually handling every refill while still allowing them to oversee the process.
"By enabling clinical staff to queue prescriptions in a compliant, role-based workflow, providers can stay focused on patient care while maintaining complete oversight." - Hannah Abrams, Product Marketing Manager, Tebra [14]
For Electronic Prescribing of Controlled Substances (EPCS), stricter protocols are required. Providers must undergo identity verification and use Multi-Factor Authentication (MFA) - such as biometrics or hardware tokens - before sending any controlled substance prescriptions [18][20].
Integrated "Pending Orders" dashboards help providers review and approve delegated tasks efficiently. During the queuing process, real-time alerts for allergies, duplicate therapies, and drug interactions should activate - no matter which staff member entered the data - ensuring potential issues are caught early [14][1].
Data Accuracy and Quality Control
Maintaining high-quality data in e-prescribing is essential for patient safety. Even with connected systems, poor data quality can lead to significant risks.
Small errors can have serious consequences. For instance, about 10.1% of ambulatory e-prescriptions include at least one issue that could confuse pharmacists or delay treatment [23]. Medication errors contribute to 7,000 to 8,000 deaths annually in the United States [19].
Data Entry Errors
Features like drop-down menus and auto-fill, designed to speed up prescribing, can sometimes introduce mistakes.
A common issue occurs when prescribers mistakenly select the wrong drug due to similar names appearing close together or when the system suggests an incorrect but frequently used dosage [1][22].
"Sig" field, where patient instructions are entered, is particularly prone to errors. In a study analyzing 25,000 e-prescriptions, 53.6% of the quality issues involved missing doses, 17.0% lacked the administration site for topical medications, and 15.8% included administrative details in the directions field [23].
For example, a 25-year-old woman faced severe complications after a 'stop' notice failed to sync between systems. She ended up filling conflicting prescriptions - one for hydroxyurea instead of hydroxychloroquine - which caused severe leukopenia and neutropenia, requiring hospitalization [21].
"The efficiencies and safety improvements that prescribers, pharmacists and patients want suffer when e-prescriptions require clarification from the prescriber because of errors or ambiguities in drug selection, quantity, directions (Sig), prescription notes or prescriber ID." - Lisa Schwartz, Senior Director, Management Affairs, NCPA [22]
Handling Unstructured Clinical Notes
Unstructured clinical notes add another layer of complexity to data integrity.
Converting free-text notes into structured data that e-prescribing systems can process is still a significant challenge.
Providers often use shorthand like "qd", "bid", or "prn", which doesn't always translate correctly into standardized fields. In fact, there are 832 different ways to write the simple instruction, "Take 1 tablet by mouth once daily" [23].
This forces pharmacists to spend an average of 5 to 10 minutes per prescription interpreting what the prescriber meant [1].
Although modern AI tools can interpret handwriting and non-standard abbreviations with over 99% accuracy [24], many systems still rely on outdated free-text fields.
Efforts like the NCPDP SCRIPT Standard V.2012, which increased the Sig field character limit from 140 to 1,000 characters, have helped reduce issues like truncated instructions [17][25].
However, this doesn’t address the broader challenge of unstructured data.
Behavioral health providers prescribing controlled substances face even greater scrutiny, making accurate, machine-readable instructions all the more critical. Solving these issues requires robust data validation measures.
Improving Data Validation
The best way to ensure data accuracy is to minimize free-text entry. Systems should require prescribers to select drugs from standardized databases using RxNorm concept unique identifiers (RxCUIs) instead of manually typing drug names [17].
Sig-builder tools can also help by breaking down instructions into discrete elements like dose, route, and frequency, ensuring all necessary details are included before submission [23].
Prescribers should carefully review drug selection, quantity, and directions on the final confirmation screen before sending prescriptions [22].
Real-time clinical decision support alerts can further enhance accuracy by flagging drug interactions, allergies, or duplicate therapies during data entry, regardless of who is inputting the information.
Instead of relying on the "Notes" field for administrative updates, staff should use dedicated transaction messages like "Cancel Prescription" or "Change Prescription" to send clear, machine-readable instructions to pharmacies [17].
Practices that implement these advanced validation tools report 35% fewer medication errors and can reduce prescribing times by up to 80% [14].
For behavioral health providers managing complex medication plans, platforms like Opus Behavioral Health EHR integrate these features directly into patient charts, minimizing errors at every step.
Meeting Regulatory Requirements
Once technical, workflow, and data quality challenges are addressed, ensuring compliance with regulatory standards becomes a critical step.
E-prescribing systems must align with federal and state regulations, as non-compliance can lead to fines, loss of billing privileges, or even legal consequences. This is especially critical for behavioral health providers who often prescribe controlled substances.
E-Prescribing Regulation Compliance
The Drug Enforcement Administration (DEA) enforces strict guidelines for Electronic Prescribing for Controlled Substances (EPCS).
For compliance, practitioners must use software that has passed a third-party audit or a DEA-approved internal review to verify it meets necessary functional criteria [26][28].
Additionally, prescribers must complete identity proofing through a Credential Service Provider (CSP) following NIST SP 800-63-3 guidelines [31][33]. While this process once required in-person notary verification, it now allows remote proofing using financial document checks and live image comparisons [33].
Two-factor authentication (2FA) is mandatory for every controlled substance prescription. This involves combining two of three factors: something you know (like a password), something you have (like a token), or something you are (biometrics) [26].
Systems must also enforce a 10-minute inactivity timeout, requiring users to re-authenticate if idle [26][30].
The Centers for Medicare & Medicaid Services (CMS) adds another layer of regulation under the SUPPORT Act. Prescribers must electronically submit at least 70% of their Schedule II–V controlled substance prescriptions for Medicare Part D beneficiaries [27][28].
Compliance is automatically tracked through Medicare Part D claims data, eliminating the need for manual reporting. However, prescribers with 100 or fewer qualifying prescriptions annually qualify for an automatic "Small Prescriber Exception" [27][29].
"EPCS enhances patient safety through patient identity checks, medication recommendations, and timely and accurate transmission of time-sensitive prescriptions." - CMS [28]
Looking ahead, organizations must prepare for the transition to NCPDP SCRIPT version 2023011, which will become mandatory for Part D e-prescribing on January 1, 2028 [2].
Although the current version, 2017071, remains acceptable, the transition period beginning July 17, 2024, allows time to update systems.
Protecting Patient Data
After meeting regulatory requirements, safeguarding patient data becomes the next priority. HIPAA compliance is mandatory throughout the e-prescribing process.
In 2024 alone, the healthcare sector reported 1,378 security incidents, with 89% leading to confirmed data breaches [31]. These statistics underscore the need for rigorous security protocols.
Data must be encrypted both in transit (using TLS 1.2 or higher) and at rest (using AES-256 encryption) to prevent unauthorized access [31].
Cryptographic modules used for digital signatures and data transmission must comply with FIPS 140-2 or higher standards [31][33].
Digital signatures are crucial for ensuring data integrity. They bind the prescriber's identity to the prescription, and any alteration after signing invalidates the record [31].
This ensures prescriptions remain tamper-proof from the provider's office to the pharmacy.
"Inadequate authentication and access controls remain one of the leading security gaps in EHR-integrated ePrescribing workflows, particularly in smaller or mid-size practices where administrative oversight is limited." - Office of the National Coordinator for Health IT [31]
Role-Based Access Control (RBAC) restricts access to sensitive patient information based on job responsibilities.
Only authorized personnel can view specific data, and systems must maintain tamper-resistant audit trails. These audit logs should record key actions - like logins, 2FA attempts, and digital signing events - with secure timestamps and user IDs [31][32].
Federal regulations require all EPCS records to be retained for a minimum of two years, though some states may mandate longer retention periods [26][30].
Pharmacy systems must also verify digital signature integrity and retain electronic prescription records exactly as received for the required period [26][30]. Additionally, software systems must synchronize their time within 5 minutes of the official National Institute of Standards and Technology (NIST) time source [30].
Simplifying Compliance Reporting
Efficient reporting mechanisms are essential for maintaining compliance.
Automated tools can ease the administrative burden. CMS tracks electronic prescribing rates via Medicare Part D claims data, so prescribers don't need to manually report or register for compliance programs [29][28]. However, organizations should monitor their compliance status to avoid penalties.
Prescribers can use the CMS EPCS Prescriber Portal (accessible with an HCQIS/HARP account) to check their annual compliance status [29][28][27].
Each September, they should review the prior year's compliance data and address any non-compliance issues promptly [28][27].
If non-compliance occurs due to factors beyond their control, such as technical failures, prescribers can apply for a waiver during the 60-day window from mid-September to mid-November [29][28][27].
Here’s a quick overview of key exceptions and actions:
|
Exception Type |
Criteria |
Action Required |
|---|---|---|
|
Small Prescriber |
100 or fewer qualifying Part D prescriptions per year |
Automatic; no action needed [29] |
|
Declared Disaster |
Located in a government-declared disaster area |
|
|
CMS-Approved Waiver |
Circumstances beyond prescriber's control (e.g., tech issues) |
|
|
Long-Term Care (LTC) |
Prescriptions for nursing facility beneficiaries |
Keeping contact information updated is critical.
Prescribers should ensure their email and physical addresses in the Medicare Provider Enrollment, Chain, and Ownership System (PECOS) and the National Plan and Provider Enumeration System (NPPES) are current.
CMS uses these systems to send compliance notices and disaster-related updates. Addresses should be updated by mid-July each year to avoid missing important communications [29][28][27].
"A prescriber's non-compliance under the CMS EPCS Program may be considered in CMS processes for assessing potential fraud, waste, and abuse, which, in some instances, could result in a referral to law enforcement or revocation of billing privileges." - CMS [29][27]
Modern e-prescribing platforms can further simplify compliance by integrating Real-Time Prescription Benefit (RTPB) tools.
These tools provide prescribers with instant information on patient insurance coverage and more affordable alternatives at the point of care [33][2].
Early adoption of NCPDP RTPB version 13, which becomes mandatory on January 1, 2027 [2], can streamline compliance and prior authorization workflows.
For behavioral health providers, platforms like Opus Behavioral Health EHR embed these compliance features into clinical workflows, reducing administrative tasks while ensuring adherence to regulatory standards.
Getting Staff to Use the System
Even a well-designed e-prescribing system - one that meets data quality, workflow needs, and regulatory standards - can fail if staff resist using it.
If clinicians fall back on paper prescriptions, regulatory compliance and patient safety benefits are lost. To ensure successful adoption, it’s crucial to address the reasons behind staff resistance.
Why Staff Resist New Systems
One major hurdle is the belief that e-prescribing slows down patient care.
A 2024 survey revealed that 71.4% of physicians still preferred paper prescriptions, citing technical difficulties and disruptions to their workflow as key issues [34].
Many worry that electronic systems increase patient wait times, especially when glitches like network outages or slow connections occur [34][35].
Another concern is the fear of making new types of errors. While paper-based prescriptions have led to 95,000 drug errors since 2000 due to illegible handwriting [8], clinicians worry about errors unique to digital systems - like selecting the wrong drug, dose, or patient from dropdown menus [4][8]. These fears, though valid, can be addressed with thoughtful system design and a gradual rollout process.
Consider the Everett Clinic’s approach: they began with a small pilot group of six enthusiastic prescribers. This phased implementation, paired with role-specific training, helped ease resistance.
Within 24 months, 200 out of 225 prescribers had adopted the system. When transitioning to Epic in 2007, they applied lessons from this initial rollout, including customizing the interface and ensuring accurate dosage mapping [35].
Tackling these concerns early paves the way for better system usability, especially in managing alerts.
Reducing Alert Overload
Alert fatigue is another common frustration. When clinicians override more than 90% of drug interaction alerts and 77% of drug allergy alerts [37], the system’s credibility takes a hit.
Approximately 7% of electronic prescriptions trigger an alert, but nearly 99% of these involve interactions with drugs the patient is already taking [37].
"Excessive alerts can undermine safety benefits. Too many alerts are generated for unlikely events, which could lead to alert fatigue." - Thomas Isaac, MD, MBA, Investigator, Beth Israel Deaconess Medical Center [37]
A streamlined alert system can make a big difference.
At Jurong Health Campus, refining the alert system led to a 74.3% reduction in interruptive alerts and a 59% average reduction across clinical groups. This allowed clinicians to focus on the alerts that truly mattered [36].
To improve alert systems, establish a governance committee to review and adjust alerts regularly. Track the "alert-to-action" ratio to identify and refine alerts that are frequently dismissed.
Configure systems to focus on clinically significant issues and use visual design strategies, like "tallman" lettering (e.g., HydrOXYzine vs. HydrALAZINE), to reduce confusion without relying on intrusive pop-ups [8][39].
Training and Support Programs
Training is perhaps the most critical factor in driving adoption.
A lack of proper training is the strongest predictor of dissatisfaction with e-prescribing systems [38]. Physicians who receive insufficient training are 3.5 times more likely to report a poor experience [38].
In the same 2024 survey, 53.6% of physicians expressed low satisfaction with their current systems, highlighting widespread training gaps [34].
Effective training goes beyond a one-time software demo. Clinicians who receive 11 or more hours of onboarding report the highest satisfaction, with 3–5 hours being the minimum for effective training [38]. Training should be tailored to specific roles and workflows rather than offering generic overviews [38].
The "Super User" model can help bridge the gap. By designating peer champions who receive advanced training months before the system goes live, organizations can provide relatable, real-time coaching. Sutter Health used this model between 2022 and 2024, focusing on efficiency training for tasks like documentation shortcuts and inbox management. This reduced after-hours work by 14% and helped 78% of physicians save 4–5 minutes per hour, reclaiming 40–60 minutes daily [38].
On-site support during the first 1–4 weeks of implementation is invaluable. Trainers can assist clinicians during patient encounters, addressing challenges in real time.
Setting up a 24/7 command center during the first week ensures immediate resolution of technical or clinical issues. Follow-up training - short, focused sessions lasting 15–60 minutes - throughout the year can introduce new features and reinforce best practices [38]. Platforms like Opus Behavioral Health EHR can integrate these training modules directly into the system, providing contextual support exactly when and where it’s needed.
Conclusion
Integrating e-prescribing into healthcare systems is no small task. The hurdles are substantial: interoperability gaps that can leave providers without critical medication histories, workflow disruptions that slow down care delivery, data quality issues that risk preventable errors, regulatory requirements that require constant vigilance, and user adoption challenges that can undermine even the most advanced systems.
The consequences of inaction are stark. Since 2000, over 95,000 prescription drug errors have been traced back to illegible handwriting on paper prescriptions. These errors contributed to more than 1.25 million serious adverse events and approximately 175,000 deaths in 2022 due to improper medication use [3][8]. For behavioral health providers managing patients with complex medication needs, the margin for error is even slimmer.
"A prescriber who can't see a patient's full medication history is making decisions with incomplete information. That's a patient safety problem." - The PIMSY Team [7]
Improving e-prescribing systems is essential to enhance safety and efficiency.
The solutions discussed - like ensuring ONC certification, complying with USCDI v3 standards, implementing single-consent workflows under 42 CFR Part 2, refining alert systems, and prioritizing robust training programs - are key to safeguarding both patients and providers.
These measures not only address interoperability and data accuracy but also help streamline workflows and boost adoption rates.
Opus Behavioral Health EHR tackles these challenges head-on by embedding e-prescribing within a unified platform that combines EHR, CRM, and RCM capabilities.
The system offers real-time interaction alerts [4], complies with Federal and DEA standards for Electronic Prescribing for Controlled Substances (EPCS) [5], and natively supports the new 42 CFR Part 2 single-consent requirements. By consolidating clinical data, Opus empowers providers to make informed decisions while staying compliant with 2026 regulations [7].
These integrated tools directly address the core issues of interoperability, workflow optimization, data accuracy, regulatory adherence, and user adoption highlighted throughout this discussion.
FAQs
How do we fix e-prescribing interoperability between different EHRs?
To tackle the challenges of e-prescribing interoperability, using standardized data exchange protocols such as HL7 and FHIR is key.
These frameworks allow prescriptions and patient information to be shared effortlessly across different EHR systems. On top of that, incorporating virtual care tools and ensuring they work smoothly within current workflows can help make e-prescribing more efficient and simplify processes across various platforms.
What’s the fastest way to reduce e-prescribing errors and rework?
The fastest way to cut down on e-prescribing mistakes is by implementing a well-designed, easy-to-use system that simplifies workflows and prioritizes precision.
Key actions include providing thorough training, tailoring the system to specific needs, and leveraging automation. Integrating the system with EHRs, offering real-time data access, and ensuring a clear, intuitive interface can significantly lower error rates.
Educating healthcare providers on efficient workflows and best practices also plays a big role in avoiding errors and reducing the need for corrections.
What is needed to be EPCS- and HIPAA-compliant without slowing clinicians down?
To meet EPCS (Electronic Prescriptions for Controlled Substances) and HIPAA (Health Insurance Portability and Accountability Act) standards without interfering with clinicians' routines, healthcare organizations need to implement certified e-prescribing systems.
These systems should feature two-factor authentication, secure identity verification, and automated audit trails. This combination helps organizations comply with regulations while keeping clinical workflows smooth and efficient.
